DGCI gives approval to Biocon's coronavirus drug for emergency use after trial on just 30 patients
Shortpedia
Content Team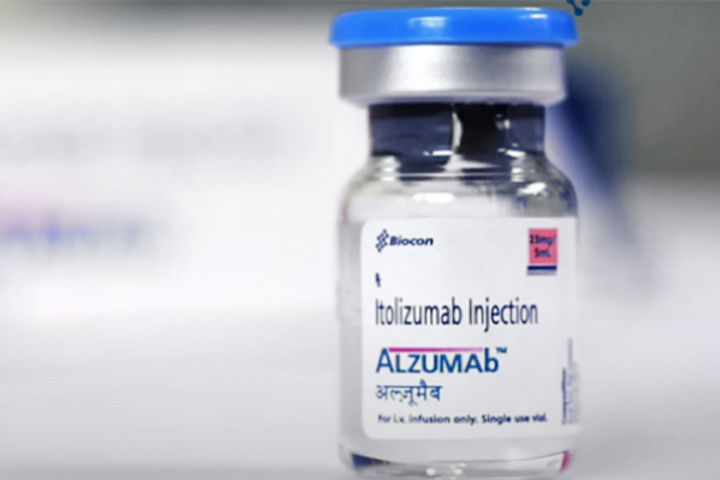
Image Credit: Shortpedia
Bengaluru-based Biocon's Itolizumab, a drug used to cure skin ailment psoriasis, has received the nod from the Drug Controller General of India (DGCI) to be used as coronavirus treatment drug for "restricted emergency use". The drug can be used on coronavirus patients with moderate to severe acute respiratory distress. It has been tested on just 30 patients. The name of the Biocon's drug is ALZUMAb.









