COVAXIN under review; may get WHO recommendation in 24 hours
Shortpedia
Content Team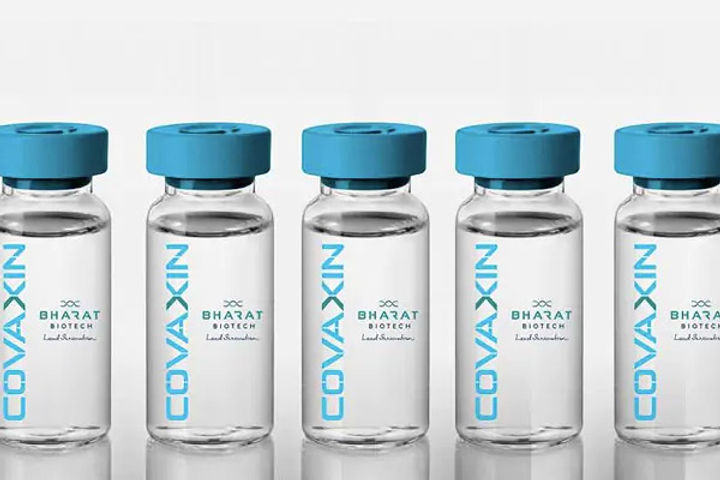
Image Credit: shortpedia
A technical advisory group of the WHO is reviewing data on COVAXIN and may recommend emergency use approval for the vaccine within 24 hours, an official said. The Indian indigenous vaccine against the coronavirus has so far been unable to secure a WHO nod due to insufficient trial data. WHO also recently said that it was seeking additional information from Bharat Biotech for a thorough evaluation of the vaccine.