Panel recommends permission for Phase I Intranasal COVID-19 vaccine trial
Shortpedia
Content Team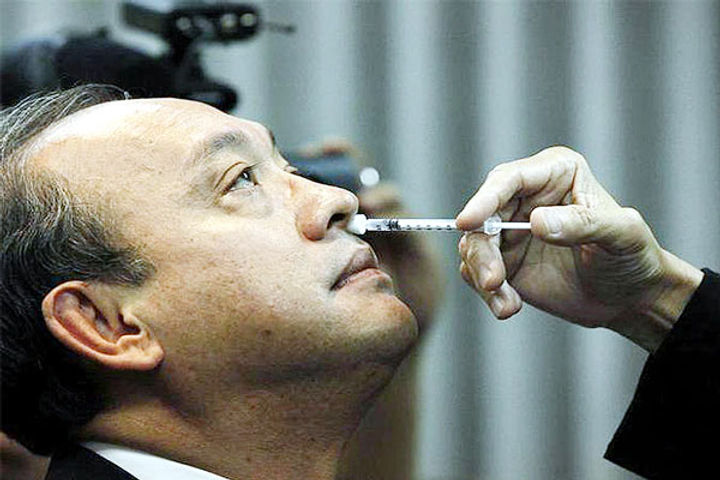
Image Credit: shortpedia
An expert panel, Central Drugs Standard Control Organisation, (CDSCO) on Tuesday recommended granting permission for conducting the Phase I clinical trial of an intranasal vaccine against COVID-19 developed by Bharat Biotech, official sources said. VK Paul, NITI Aayog member (Health) said, "If this vaccine works, it can be a game-changer because it is so easy to use and we look forward to this development."